Diadematoid echinoids: A cryptic part of the tropical fossil record
Stephen K Donovan (The Netherlands)
The writers of holiday brochures invariably fail to mention, let alone emphasise, the bad points of a location. For example, I’ve lived in both Jamaica and the Netherlands, and, for me, the thing that unites these two countries is the number of mosquitoes. However, as mosquitoes aren’t a good sales point with tourists, they are carefully ignored in holiday brochures and advertisements.
Another Caribbean critter that doesn’t get mentioned until you actually arrive and want to go for a dip in the sea is the sea urchin, known in Jamaica as a sea egg. In truth, any danger to the unwary swimmer comes from the few species of regular echinoid that have long, pointed spines. These are found in many shallow water habitats, but are best concealed (and, therefore, most dangerous to the swimmer) in seagrass beds. The best protection from these echinoids is to wear an old pair of training shoes that you’d be happy to dispose of at the end of the vacation. However, without prior knowledge, who would take such a thing on holiday with them?
Among these echinoids, the one most likely to ruin your holiday is the black, long-spined urchin, Diadema antillarum (Phillipi) in the Caribbean; which is broadly similar in morphology to the figured specimen, Diadema setosum (Leske) from Indonesia (Fig. 1). These echinoids have relatively small bodies, but numerous long, needle-like spines. These are, essentially, single calcite crystals. The unsuspecting swimmer treading on such an urchin will have their foot pierced by many needle-like spines that break off along calcite cleavage planes with the slightest movement. The urchin has efficiently protected itself, but one more tropical holiday has been ruined.

Diadema antillarum are widespread in the Caribbean region, where they are among the commonest and most prominent echinoids in shallow water environments. Yet, it is peculiar to relate that their fossil record was, until recently, apparently poor. Until about 20 years ago, D. antillarum and, indeed, all related diadematoid echinoids, had no known fossil record in the Antilles. What was not known was the meaning of the pattern – was it a preservational artefact or did it indicate that D. antillarum and related species were recent migrants into the Caribbean region?
The answer to this conundrum was provided in impressive style by my former research student, Carla Gordon, as part of her studies at the University of the West Indies in Kingston, Jamaica. Carla was researching the fossil echinoids of the Late Pleistocene Falmouth Formation for her MPhil thesis (Gordon, 1991). Unfortunately, echinoid tests are rare in this deposit (exceptions were described by Donovan & Collins, 1997; Simpson, 2001) and Carla’s research was based almost entirely on disarticulated ossicles. These were mainly spines and test plates, with rarer elements of Aristotle’s lantern sieved from bulk samples or, in the case of some large, obvious fragments, taken from coastal exposures (Fig. 2).

Over 10,000 separate echinoid ossicles were picked from samples, which was a magnificent effort. The majority were broken fragments of spines of diadematoid echinoids. This was an extraordinary discovery – it changed the fossil record of diadematoids in the region from a total absence to, quite suddenly, many thousands of specimens. Admittedly, they were all fragments, but this was Carla’s innovative approach yielding abundant results. She was the first Antillean palaeontologist to apply micropalaeontological techniques to the study of echinoids. This research project showed that about half of the shallow water echinoid taxa known from Jamaica today (Hendler et al., 1995, pp. 197-249) also occur in the Late Pleistocene Falmouth Formation raised reef (Gordon & Donovan, 1992; Donovan & Gordon, 1993).
Carla’s specimens were most probably derived from D. antillarum (Table 1).

There are other diadematoids known from the shallow water of the Caribbean today. Diadema antillarum itself is known from 0-400m water depth; Astropyga magnifica A.H. Clark occurs at 11 to 88m; and Centrostephanus longispinus (Philippi) lives at 33 to 310m (Serafy, 1979; Hendler et al., 1995). These species can be differentiated on the basis of their gross morphology, but also from their spines (Fig. 3). Comparison of Carla’s spines with those in Fig. 3 indicates that they are D. antillarum. This assessment agrees well with palaeoenvironmental determinations of this site as a patch reef in the lagoon behind the main barrier reef crest (Fig. 2). For example, the scleractinian coral species found here are typically from shallow waters (Liddell et al., 1984, p. 77, fig. 46, table 9).

The recognition of fossil diadematoids from the Late Pleistocene of Jamaica showed where and how to look for them elsewhere (Table 1). Therefore, diadematoid spines have now been recognised from Pleistocene raised reefs on other islands, such as Tobago (Donovan et al., 2001) and, from the pre-Pleistocene, the oldest currently known is Early Palaeocene (Danian) (Donovan & Veltkamp, 1992; Fig. 4). So, the apparent lack of a fossil record of diadematoids in the Antilles was, indeed, an artefact of our prevalent collecting methods. By pursuing the micro rather than macropalaeontological record, diadematoids were found to be present, even locally common, in a sedimentary record spanning over 60Ma.

However, the Caribbean was not the only tropical region from which diadematoids were unexpectedly unknown from the fossil record (Jeannet & Martin, 1937). In Southeast Asia, extant shallow water diadematoids are, like the Antilles, common if not particularly speciose (Miskelly, 2002; Schultz, 2005). But, like the Antilles, in studies of fossil echinoids from Southeast Asia, the diadematoids had been noticeably absent. This lack tantalised both my colleague, Dr Willem Renema, and me. Dr Renema has a particular research interest in the fossil and extant larger benthic foraminifera of Southeast Asia (see, for example, Renema, 2002).
As a micropaleontologist, Willem and his students were (and are) actively picking sediment samples from the Cenozoic of the region, and continue to find many unusual specimens, including fragments of echinoderms, particularly echinoids. To cut a long story short, diadematoids were there as fragments (Donovan et al., 2012), similar to those of the Antilles (compare Fig. 5 with Figs. 3 and 4). They appear to be less common than from the Falmouth Formation, although other Antillean localities are similarly depauperate in specimens.
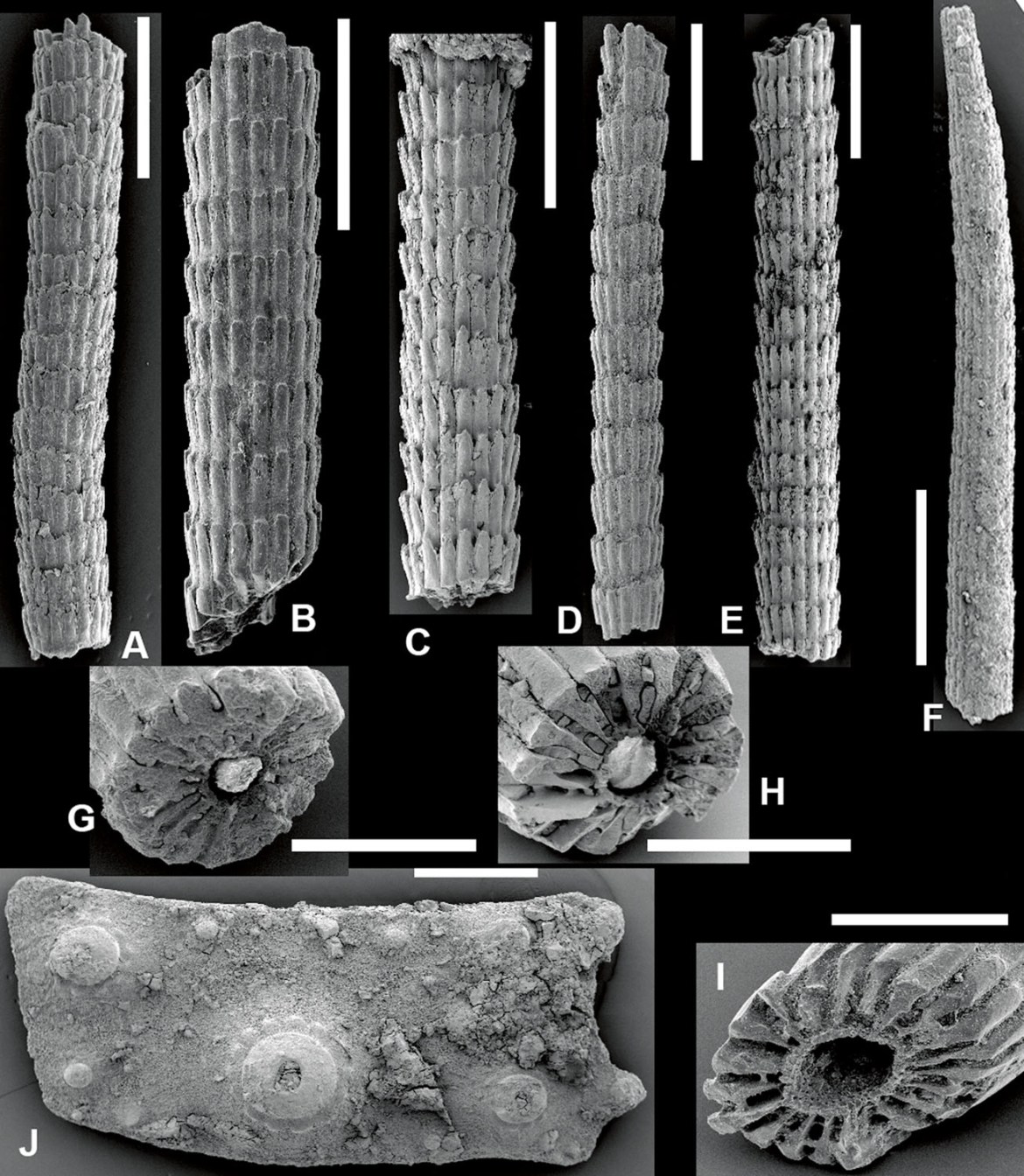
Donovan et al. (2012) is the first report of fossil diadematoid echinoids from reef-related (or any) rocks in Indonesia. Geologists studying Cenozoic reefs are mainly concerned with those organisms that have left a robust skeleton, such as scleractinian corals. It is widely recognised that some reef organisms, common today, are rare in these ancient deposits because they disarticulate rapidly after death and, therefore, do not form a prominent fossil component, such as sponges. Diadematoids are analogous.
They are common in modern reef environments, but almost completely unknown from Neogene fossil reefs. This absence can be interpreted as real (diadematoids only invaded reef environments in the Holocene) or an artefact (the poor preservational potential of diadematoids did not favour their identification in ancient reefs). The latter position – that diadematoids were a component of the Neogene reef biota in the Antilles and Indonesia – is now known to be the correct interpretation.
References
Donovan, S.K. 2005. The fossil record of Diadema in the Caribbean. Coral Reefs, 24: 603-605.
Donovan, S.K. & Collins, J.S.H. 1997. Unique preservation of an Echinometra Gray (Echinodermata: Echinoidea) in the Pleistocene of Jamaica. Caribbean Journal of Science, 33: 123‑124.
Donovan, S.K. & Gordon, C.M. 1993. Echinoid taphonomy and the fossil record: supporting evidence from the Plio‑Pleistocene of the Caribbean. Palaios, 8: 304‑306.
Donovan, S.K. & Jackson, T.A. (in press). Jamaica’s geodiversity (Part 2): highlights from the Neogene. Deposits.
Donovan, S.K., Lewis, D.N., Vincent, H. & Blackman, I.M. 2001. Pleistocene echinoids from Tobago, West Indies. Caribbean Journal of Earth Science, 35: 1-12.
Donovan, S.K. & Paul, C.R.C. 1998. Echinoderms of the Pliocene Bowden shell bed, southeast Jamaica. Contributions to Tertiary and Quaternary Geology, 35: 129-146.
Donovan, S.K., Renema, W., Pinnington, C.A. & Veltkamp, C.J. 2012 (in press). Significance of diadematid echinoid ossicles in micropalaeontological samples, Miocene-Pliocene of Indonesia. Alcheringa.
Donovan, S.K. & Veltkamp, C.J. 1992. A diadematoid echinoid from the Lower Paleocene of Jamaica. Caribbean Journal of Science, 28: 222‑224.
Gordon, C.M. 1990. Taxonomy and Palaeoecology of the Echinoids of the Late Pleistocene Falmouth Formation of Jamaica. University of the West Indies, Mona: xvi+297 pp.
Gordon, C.M. & Donovan, S.K. 1992. Disarticulated echinoid ossicles in paleoecology and taphonomy: the last interglacial Falmouth Formation of Jamaica. Palaios, 7: 157‑166.
Gordon, C.M. & Donovan, S.K. 1994. Some fossil echinoids (Echinodermata) from the Neogene of St. Croix, U.S. Virgin Islands. Caribbean Journal of Science, 30: 69‑75.
Hendler, G., Miller, J.E., Pawson, D.L. & Kier, P.M. 1995. Sea Stars, sea Urchins and Allies: Echinoderms of Florida and the Caribbean. Smithsonian Institution Press, Washington, D.C.: xi+390 pp.
Jeannet, A. & Martin, R. 1937. Ueber Neozoische Echinoidea aus dem Niederlaendisch-Indischen archipel. Overdruk uit Leidsche Geologische Mededeelingen, 8: 215–308.
Kroh, A. 2005. Echinoidea Neogenica. Catalogus Fossilium Austriae, 2: lvi+210 pp.
Liddell, W.D., Ohlhorst, S.L. & Coates, A.G. 1984. Modern and Ancient Carbonate Environments of Jamaica. Sedimenta, 10: vii+98 pp.
Miskelly, A. 2002. Sea Urchins of Australia and the Indo-Pacific. Capricornica Publications, Sydney: 179 pp.
Renema, W. 2002. Larger Foraminifera as Marine Environmental Indicators. Scripta Geologica, 124: 1-263.
Simpson, G.A. 2001. First record of the holectypoid echinoid Echinoneus cyclostomus Leske from the late Pleistocene Falmouth Formation of Jamaica. Caribbean Journal of Earth Science, 35 (for 2000): 33-36.
Schultz, H. 2005. Sea Urchins: A Guide to worldwide Shallow Water Species. Heinke & Peter Schultze Partner, Hemdingen: xii+484 pp.

